They're not in the business for only altruistic reasons......Besides that, if you get COVID from being inside a business - like a restaurant or WalMart - they're immune from liability, at least here in Georgia, anyway. So why shouldn't Pfizer get the same treatment? They're trying to fix the problem, not just sell more fries.
Colleges
- AAC
- ACC
- Big 12
- Big East
- Big Ten
- Pac-12
- SEC
- Atlantic 10
- Conference USA
- Independents
- Junior College
- Mountain West
- Sun Belt
- MAC
- More
- Navy
- UAB
- Tulsa
- UTSA
- Charlotte
- Florida Atlantic
- Temple
- Rice
- East Carolina
- USF
- SMU
- North Texas
- Tulane
- Memphis
- Miami
- Louisville
- Virginia
- Syracuse
- Wake Forest
- Duke
- Boston College
- Virginia Tech
- Georgia Tech
- Pittsburgh
- North Carolina
- North Carolina State
- Clemson
- Florida State
- Cincinnati
- BYU
- Houston
- Iowa State
- Kansas State
- Kansas
- Texas
- Oklahoma State
- TCU
- Texas Tech
- Baylor
- Oklahoma
- UCF
- West Virginia
- Wisconsin
- Penn State
- Ohio State
- Purdue
- Minnesota
- Iowa
- Nebraska
- Illinois
- Indiana
- Rutgers
- Michigan State
- Maryland
- Michigan
- Northwestern
- Arizona State
- Oregon State
- UCLA
- Colorado
- Stanford
- Oregon
- Arizona
- California
- Washington
- USC
- Utah
- Washington State
- Texas A&M
- Auburn
- Mississippi State
- Kentucky
- South Carolina
- Arkansas
- Florida
- Missouri
- Ole Miss
- Alabama
- LSU
- Georgia
- Vanderbilt
- Tennessee
- Louisiana Tech
- New Mexico State
- Middle Tennessee
- Western Kentucky
- UTEP
- Florida International University
High School
- West
- Midwest
- Northeast
- Southeast
- Other
- Alaska
- Arizona
- California
- Colorado
- Nevada
- New Mexico
- Northern California
- Oregon
- Southern California Preps
- Washington
- Edgy Tim
- Indiana
- Kansas
- Nebraska
- Iowa
- Michigan
- Minnesota
- Missouri
- Oklahoma Varsity
- Texas Basketball
- Texas
- Wisconsin
- Delaware
- Maryland
- New Jersey Basketball
- New Jersey
- New York City Basketball
- Ohio
- Pennsylvania
- Greater Cincinnati
- Virginia
- West Virginia Preps
ADVERTISEMENT
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Vaccine Shaming
- Thread starter MikeHoncho69
- Start date
Sounds like you’re ready for a career change.I’ve taken their vaccine. I am not moving any posts.
I know people that have taken ivermectin and overcome Covid quickly. There’s one little problem. It’s cheap and readily available. Almost all docs work for Big Hospital Corps. Big Pharma and Big Hospitals make no money with ivermectin working.
A highly educated population so surprised to see much pushback although Zville does take in a fair number of students from the blue collar area of Whitestown.Yep. Zionsville voted last night to mask all children under 12. They had quite a bit of pushback at that meeting as well. People are fed up.
I'll refrain from the obvious horse joke . . . .I’ve taken their vaccine. I am not moving any posts.
I know people that have taken ivermectin and overcome Covid quickly. There’s one little problem. It’s cheap and readily available. Almost all docs work for Big Hospital Corps. Big Pharma and Big Hospitals make no money with ivermectin working.
kind of. the immunity is only four years and the case would sound in product liability where many states hold five year sols.Pfizer is still immune from liability when their vaccine injures someone so that approval isn't worth the paper it's written on.
You need to learn the reasons behind the limited release from liability.Pfizer is still immune from liability when their vaccine injures someone so that approval isn't worth the paper it's written on.
Case study:
Bendectin, an antinausea drug that really does eliminate morning sickness in pregnant women, and that (unlike phalidomide) has no effects on the fetus. It was approved to market in the 1970s. 1 in every 33 babies born in the USA, with mothers taking no drugs at all, unfortunately is born with a birth defect. 1 in every 33 babies born in the USA, with mothers taking Bendectin, unfortunately was born with a birth defect. The drug clearly didn't cause or prevent birth defects. But if you took the drug and had a birth defect, you blamed the drug. Lawsuit after lawsuit after lawsuit. More than 10. All won by the pharma company, since the data was crystal clear that the drug was not causing any of them.
After spending millions to defend (and win!) every lawsuit, and taking PR blows all along, the pharma company said "this crap ain't worth it. Let's pull the drug off the market, and let's never EVER EVER work on women's health or pediatric drugs (including vaccines) again".
What are the consequences? No drug discovery for portions of the population that need it.
The remedy? The government said that we will shield you from some liability (throwing out nuisance lawsuits) if you will please work in that area. You are not sheilded from liability due to fraud, deceit, negligence, false claims, or anything intentional on your part. You will be taken to the cleaners is you do that.
It is the price to pay to make it even possible for drug companies to work in drug discovery for those highly litigious populations.
Lmao no. I have no clue what year you're talking about but Pfizer is projected to profit 35 billion this year from the vax. And there are no nuisance suits. These cases are expensive as shit to bring. That's why most are referred to a group of deep pocket firms and they end up in mdls. Type of cases you spend on and hope to get paid many years laterYou need to learn the reasons behind the limited release from liability.
Case study:
Bendectin, an antinausea drug that really does eliminate morning sickness in pregnant women, and that (unlike phalidomide) has no effects on the fetus. It was approved to market in the 1970s. 1 in every 33 babies born in the USA, with mothers taking no drugs at all, unfortunately is born with a birth defect. 1 in every 33 babies born in the USA, with mothers taking Bendectin, unfortunately was born with a birth defect. The drug clearly didn't cause or prevent birth defects. But if you took the drug and had a birth defect, you blamed the drug. Lawsuit after lawsuit after lawsuit. More than 10. All won by the pharma company, since the data was crystal clear that the drug was not causing any of them.
After spending millions to defend (and win!) every lawsuit, and taking PR blows all along, the pharma company said "this crap ain't worth it. Let's pull the drug off the market, and let's never EVER EVER work on women's health or pediatric drugs (including vaccines) again".
What are the consequences? No drug discovery for portions of the population that need it.
The remedy? The government said that we will shield you from some liability (throwing out nuisance lawsuits) if you will please work in that area. You are not sheilded from liability due to fraud, deceit, negligence, false claims, or anything intentional on your part. You will be taken to the cleaners is you do that.
It is the price to pay to make it even possible for drug companies to work in drug discovery for those highly litigious populations.
Last edited:
There's also one big problem. Several controlled clinical trials for ivermectin were approved and were run. The FDA and NIH poured a lot of money into it, actually, since it obviously would have been a cost-effective route.I know people that have taken ivermectin and overcome Covid quickly. There’s one little problem. It’s cheap and readily available. Almost all docs work for Big Hospital Corps. Big Pharma and Big Hospitals make no money with ivermectin working.
All of the blinded, controlled clinical trials showed that it does not work. At all.
So on one side we have the gold standard, the double blinded controlled clinical trial results.
On the other side we have a few doctors using it off label who say that they gave it to 15 patients and 14 recovered, when before they gave nothing to 15 patients and "only" 13 recovered. They can swear up and down that they "think" ivermnectin made the difference, but it does not make it so.
I guess we will see if these folks get better.There's also one big problem. Several controlled clinical trials for ivermectin were approved and were run. The FDA and NIH poured a lot of money into it, actually, since it obviously would have been a cost-effective route.
All of the blinded, controlled clinical trials showed that it does not work. At all.
So on one side we have the gold standard, the double blinded controlled clinical trial results.
On the other side we have a few doctors using it off label who say that they gave it to 15 patients and 14 recovered, when before they gave nothing to 15 patients and "only" 13 recovered. They can swear up and down that they "think" ivermnectin made the difference, but it does not make it so.
Edit: @outside shooter

Ivermectin Wins in Court Again: For Human Rights
One dose of Ivermectin was all it took to get 81-year-old John Swanson off the ventilator. John’s wife Sandra could not believe it. His story is remarkably similar to other
Last edited:
If you remember I had private message with you:Lmao no. I have no clue what year you're talking about but Pfizer is projected to profit 35 billion this year from the vax. And there are no nuisance suits. These cases are expensive as shit to bring. Heavy bucks to bring. That's why most are referred to a group of deep pocket firms and they end up in mdls. Type of cases you spend on and hope to get paid many years later
My brother in law had a BHR Birmingham Hip Resurfacing.
The replacement started chipping or whatever and he ended up with Cobalt Poisoning from the implant. He’s had terrible pain all over his body for some time. They just discovered the poisoning
A week ago they replaced one. There’s still a second to replace.
My sister called the firm you gave me that won a class action suit regarding this poisoning. Turned out my brother in laws serial number wasn’t one on the settlement list. They weren’t interested in his case. Point being, you’re right, these cases aren’t easy.
Last edited:
Pfizer is still immune from liability when their vaccine injures someone so that approval isn't worth the paper it's written on.
Such a bummer. Keep tracking it tho. Things may change. But yes they are exceedingly expensive to bring and take forever with bellwether trials etcIf you remember I had private message with you:
My brother in law had a BHR Birmingham Hip Resurfacing.
The replacement started chipping or whatever and he ended up with Cobalt Poisoning from the implant. He’s had terrible pain all over his body for some time. They just discovered the poisoning
A week ago they replaced one. There’s still a second to replace.
My sister called the firm you gave me that won a class action suit regarding this poisoning. Turned out my brother in laws serial number wasn’t one on the settlement list. They weren’t interested in his case. Point being, you’re right, these cases aren’t easy.
I guess we will see if these folks get better.
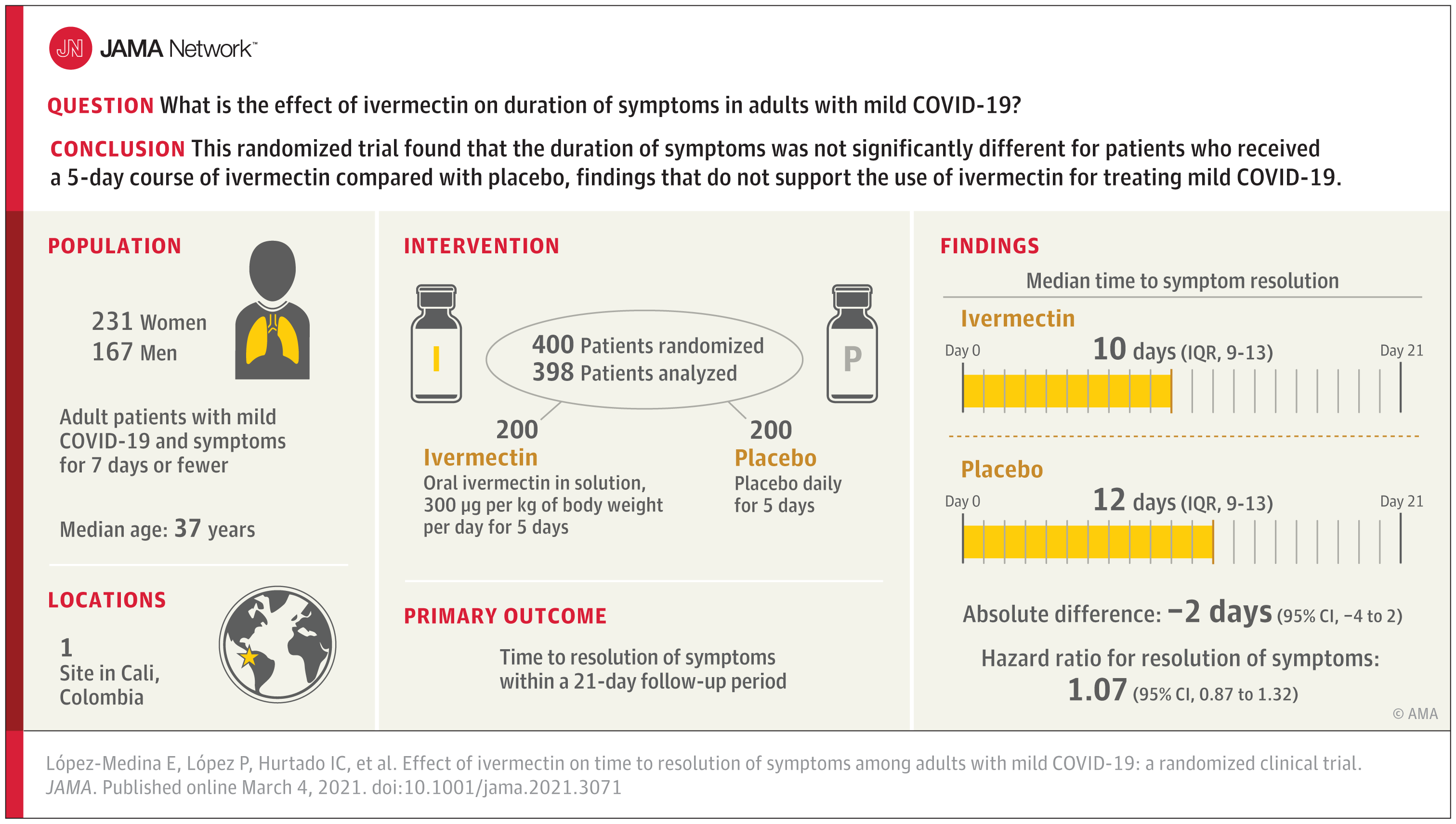
Effect of Ivermectin on Time to Resolution of Symptoms Among Adults With Mild COVID-19
This randomized trial compares the effects of ivermectin vs placebo on time to symptom resolution within 21 days among patients with mild COVID-19.
Among 400 adults with mild COVID-19, a 5-day course of ivermectin, compared with placebo, did not significantly improve the time to resolution of symptoms. The findings do not support the use of ivermectin for treatment of mild COVID-19

Ivermectin as a potential treatment for mild to moderate COVID-19 – A double blind randomized placebo-controlled trial
Objective Ivermectin has been suggested as a treatment for COVID-19.This randomised control trial was conducted to test the efficacy of Ivermectin in the treatment of mild and moderate COVID-19. Design Parallel, double blind, randomised, placebo controlled trial Setting: A tertiary care...

Efficacy and safety of Ivermectin and Hydroxychloroquine in patients with severe COVID-19. A randomized controlled trial
Background In the search for active drugs against COVID-19, the indications of many have been redirected. Ivermectin and Hydroxychloroquine are drugs that inhibit viral replication in vitro and that have been used in several medical centers. Objectives This clinical trial analyzes the efficacy...
(there are many other such reports)
one somewhat conflicting report:

Effectiveness of Ivermectin as add-on Therapy in COVID-19 Management (Pilot Trial)
Background To date no effective therapy has been demonstrated for COVID-19. In vitro, studies indicated that ivermectin (IVM) has antiviral effect. Objectives To assess the effectiveness of ivermectin (IVM) as add-on therapy to hydroxychloroquine (HCQ) and azithromycin (AZT) in treatment of...
Similar threads
- Replies
- 16
- Views
- 308
- Replies
- 19
- Views
- 452
- Replies
- 0
- Views
- 109
ADVERTISEMENT
ADVERTISEMENT

